Carbon, the Miracle Element
A lot of people today worry about the carbon footprints of nations, industries, technologies, and even individual human beings and animals. They worry that our activities are pumping too much carbon in the form of carbon dioxide and methane— both powerful greenhouse gases—into the atmosphere and too much carbon in the form of tiny particles of soot onto glaciers and snowfields. They worry that these activities are warming our planet and raising our sea levels.
However, the fact that carbon exists at all is nothing short of a miracle. And for Earth to have been blessed with neither too much carbon nor too little carbon for advanced life to exist is another miracle. And the fact that Earth has its stores of carbon distributed into the just right locations for human civilization to thrive ranks as yet another miracle of divine design.
Is There a Carbon Footprint Problem?
In the context of the latter miracle, the concerns about our carbon footprints are justified. In the last 250 years we humans have moved vast quantities of carbon in the form of fossil fuels, forests, farms, and farm animals into atmospheric carbon dioxide, methane, and particulates. Atmospheric carbon dioxide levels before the industrial revolution were 280 parts per million by volume.1 Today, they measure 408 parts per million.2 Carbon particles are black. When they fall on glaciers and snowfields they cause heat from the Sun to be absorbed by glaciers and snowfields rather than being reflected away.
Our carbon footprints are melting away the Arctic ice cap. Figure 1 shows the perennial Arctic ice cap as it was in 1980. Figure 2 shows it as it is today.
Figure 1: Extent of Perennial Arctic Sea Ice in 1980. Image credit: NASA
Figure 2: Extent of Perennial Arctic Sea Ice in 2016. Image credit: NASA
Paradoxically, the melting of the polar ice cap could lower the sea level and cool the planet. Open ocean water absorbs more heat from the Sun than does ice. That extra heat absorption could cause more Arctic Ocean water to evaporate, which could result in much more snow falling on Canada and Siberia. That extra snow could lead to the formation of icefields and glaciers in Canada and Siberia, which would lower the sea level, cool the Northern Hemisphere, and even bring on the next ice age.
What we do with Earth’s stores of carbon can dramatically impact Earth’s climate, geology, wildlife, and our civilization. When God told us to manage Earth’s resources for our benefit and of all Earth’s life (Genesis 1:28–30; Job 37–39), that management included wise stewardship of the carbon resources with which God blessed our planet.
Cosmic Mass Fine-Tuned for Carbon
One of the more frequent challenges I get from nontheists is the following: “If there is a God who wanted to create a home for human beings, why would he create hundreds of billions of useless galaxies?” The quick answer is that, given the laws of physics God chose for the universe, it is not possible to make a planet on which humans can live and thrive without the hundreds of billions of galaxies. In fact, it is not possible for any kind of physical life to exist without hundreds of billions of galaxies.
The universe’s population of galaxies makes up much of the cosmic mass density. For physical life to be possible, the cosmic mass density must be fine-tuned.
The universe begins with only one element, hydrogen. The cosmic mass density determines how much of the universe’s primordial hydrogen gets fused into helium during the first few minutes of cosmic history.
As the universe expands from the cosmic creation event, it cools from a near infinitely high temperature down to its present mean temperature of about 2.725° Celsius above absolute zero (-273.15° Celsius). There is a brief episode when the universe passes through the temperature range (150–15 million degrees Celsius) at which hydrogen fusion occurs.
If the cosmic mass density is the slightest bit smaller than what it is, so little helium is fused from hydrogen during the first several minutes of cosmic history that the nuclear furnaces of future stars are not able to make any elements heavier than helium. In that event, the periodic table of elements never advances beyond hydrogen and helium (see figure 3).

Figure 3: The Periodic Table of Elements for a Slightly Less Massive Universe.
If the cosmic mass density is the slightest bit larger than what it is, so much helium is fused from hydrogen during the first several minutes of cosmic history that the nuclear furnaces of future stars quickly convert all the baryonic matter (matter comprised of protons, neutrons, and electrons) into elements heavier than iron. In that event, the periodic table of elements contains no elements lighter than cobalt (see figure 4).
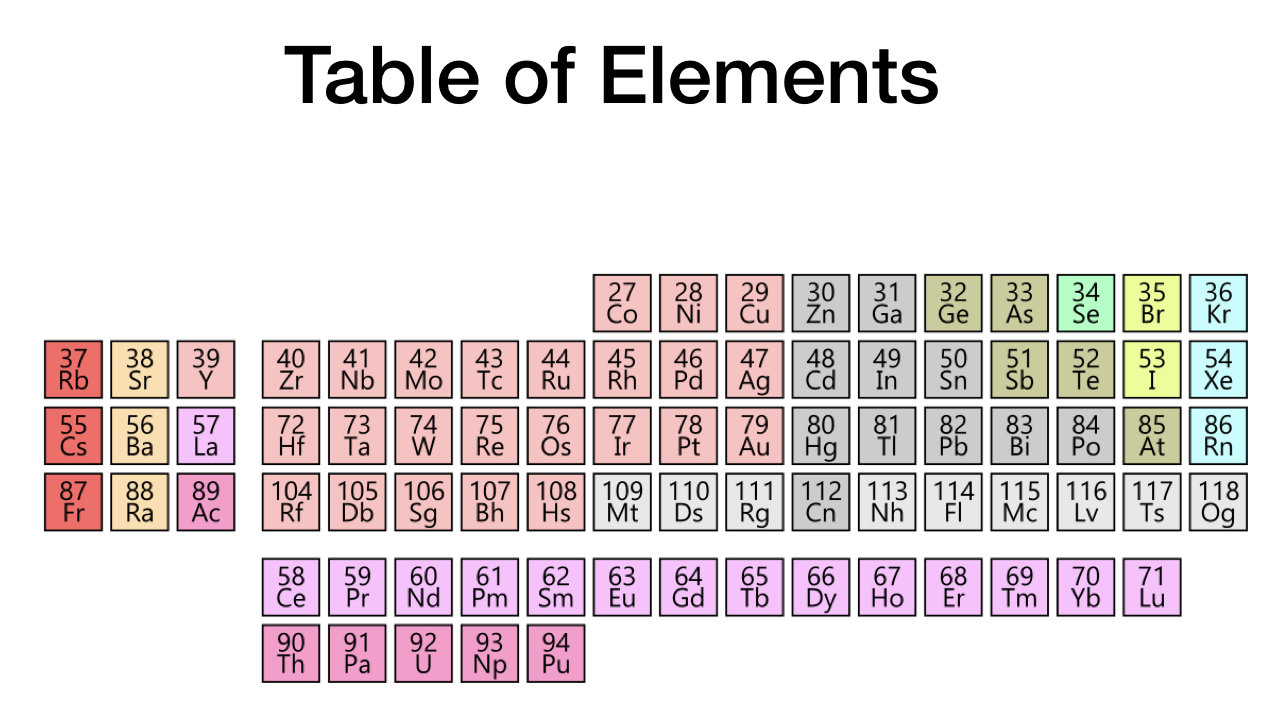
Figure 4: The Periodic Table of Elements for a Slightly More Massive Universe.
The periodic tables for both a slightly less massive universe and slightly more massive universe are missing the most life-critical elements: carbon, nitrogen, oxygen, sodium, magnesium, phosphorus, sulfur, chlorine, potassium, calcium, vanadium, chromium, and manganese. Without carbon, no conceivable kind of physical life is possible. Unless the universe possesses precisely the mass that it does, carbon will be missing—and so will life. It is a miracle of cosmic design that the universe contains any carbon at all.
Nuclear Resonance Levels Fine-Tuned for Carbon
Not only must the cosmic mass density be exquisitely fine-tuned for the universe to contain carbon, so must the nuclear resonance levels for helium, beryllium, carbon, and oxygen. Stars fuse carbon and oxygen from helium through the triple-alpha process. In this process two helium nuclei fuse together to make beryllium. Then, a helium nucleus fuses with a beryllium nucleus to make carbon. Then, some carbon nuclei fuse with helium nuclei to make oxygen.
The only reason that the triple-alpha process produces any carbon or oxygen at all is because in the first step, the ground state energy level of the beryllium-8 nucleus almost exactly equals the ground state energy level of two helium-4 nuclei. In the second step, the ground state energy level of a beryllium-8 nucleus plus a helium-4 nucleus almost exactly equals the energy level of an excited state of a carbon-12 nucleus. In the third step, the ground state energy level of a carbon-12 nucleus at 7.65 million electron volts is slightly larger than the ground state energy level of an oxygen-16 nucleus at 7.12 million electron volts.3
If it were not for the near equivalences or resonances of the nuclear energy levels of two helium nuclei relative to a beryllium nucleus and a beryllium nucleus plus a helium nucleus relative to a carbon nucleus, the universe would contain very little or no carbon and very little or no elements heavier than carbon. Life would be impossible. Furthermore, unless the difference in the nuclear energy levels between a carbon nucleus and an oxygen nucleus was precisely 0.53 million electron volts, the universe either would contain a lot of carbon and no oxygen or a lot of oxygen and no carbon. Either way, physical life would be impossible in the universe.
Astronomer Fred Hoyle and physicist Willy Fowler in the early 1950s were the first to understand how critical the relative nuclear energy levels of helium, beryllium, carbon, and oxygen were for making life possible in the universe. Commenting on the highly fine-tuned nature of these nuclear energy levels, Hoyle wrote in an article he published in Engineering & Science,4
A common sense interpretation of the facts suggests that a super intellect has monkeyed with the physics, as well as with chemistry and biology, and that there are no blind forces worth speaking about in nature. The numbers one calculates from the facts seem to me so overwhelming as to put this conclusion beyond question.
Removal of Volatiles Fine-Tuned for Carbon
Without carbon, life is impossible. However, unless the quantity of carbon in and on a planet is fine-tuned, life, physical life, and certainly advanced physical life will not be possible. A planet lacking in carbon will not have sufficient carbon for life chemistry to function. A planet with too much carbon will possess a life-suffocating atmosphere.
Carbon is ubiquitous in the universe. There are no carbon-free bodies in the universe. There are, however, a great many carbon-rich bodies. In fact, carbon-rich bodies are the norm.
As I described in my book, Improbable Planet, extrasolar planets that are the most similar to Earth, where astronomers are able to measure, or at least estimate, the carbon abundance of these planets, possess about 1,200 times as much carbon-based atmospheric gas as Earth.5 This much carbon-based atmospheric gas would rule out photosynthetic life, all animals, and likely all physical life.
How did Earth become so extremely carbon-poor? The answer is that it was born in a very different location from where it presently resides. Earth’s birth occurred in a dense cluster of more than 10,000 stars (see figure 5) located much closer to the center of the Milky Way Galaxy. There, the primordial Earth was exposed to several nearby supernovae and to the winds of several nearby Wolf-Rayet stars. Consequently, the primordial Earth was bathed in huge quantities of radiation from the radiometric decay of aluminum-26.6

Figure 5: The solar system’s birthplace was in a star cluster similar to this one. Image credit: European Southern Observatory
Earth’s Perfect Carbon Balance
The radiation from the decay of aluminum-26 blasted away most of the volatiles (gas and liquids) that the primordial Earth possessed. The Moon-forming event (see figure 6) which occurred a few tens of millions of years later removed all or virtually all of the volatiles that remained.7 Later, comets restored a tiny fraction of the volatiles that Earth had lost.8
Figure 6: Artist’s Depiction of the Collision between the Primordial Earth and Another Planetary Body That Formed the Moon. Image credit: NASA
Earth’s unique early history resulted in its possessing exactly the right amount of carbon to optimally sustain advanced life. The carbon it does possess is optimally distributed throughout Earth’s environment to optimally benefit humans and human civilization. In studying the universe’s carbon and Earth’s carbon, we become witnesses to multiple miracles of divine design for the benefit of physical life and especially for the benefit of human beings and human civilization.
Endnotes
- D. M. Etheridge et al., “Natural and Anthropogenic Changes in Atmospheric CO2 over the Last 1000 Years from Air in Antarctic Ice and Firn,” Journal of Geophysical Research 101 (February 20, 1996): 4115–28, doi:10.1029/95JD03410.
- National Oceanic and Atmospheric Administration, Earth System Research Laboratory, Global Monitoring Division, “Trends in Atmospheric Carbon Dioxide: Recent Monthly Average Mauna Loa CO2,” January 2018, https://www.esrl.noaa.gov/gmd/ccgg/trends/.
- Fred Hoyle, “The Universe: Past and Present Reflections,” Engineering & Science (November 1981): 12, https://calteches.library.caltech.edu/3312/1/Hoyle.pdf.
- Hoyle, “The Universe.”
- Hugh Ross, Improbable Planet: How Earth Became Humanity’s Home (Grand Rapids: Baker, 2016), 35–36.
- J. D. Gilmour and C. A. Middleton, “Anthropic Selection of a Solar System with a High 26Al/27Al Ratio: Implications and a Possible Mechanism,” Icarus 201 (June 2009): 821–23, doi:10.1016/j.icarus.2009.03.013.
- Ross, Improbable Planet, 48–58; Hugh Ross, “Yet More Reasons to Thank God for the Moon,” Today’s New Reason to Believe (blog), Reasons to Believe, November 22, 2016, /todays-new-reason-to-believe/read/todays-new-reason-to-believe/2016/11/22/yet-more-reasons-to-thank-god-for-the-moon.
- Ross, Improbable Planet, 57–60.