Most Extreme Extremophile
All of us can remember at least one really strange friend, teacher, relative, or associate. Among Earth’s species, too, are many really strange life-forms. Biologists call them extremophiles—life that can’t live under benign conditions but are able to survive and, to some limited extent, even thrive under hostile conditions. A list of well-studied extremophiles includes (-phile = loving; -tolerant = able to put up with):
- thermophiles: survive at temperatures between 120° and 160° Fahrenheit
- hyperthermophiles: survive at temperatures between 175° and 235° Fahrenheit
- psychrophiles: survive at temperatures between 25° and 39° Fahrenheit
- acidophiles: survive under high acidity conditions
- alkalophiles: survive under extreme alkaline conditions
- halophiles: survive in environments containing 20–30 percent salt
- barophiles: survive at pressures 300–700 times sea level air pressure
- radiotolerants: can survive a high radioactive environment
- xerophiles: can survive in extremely dry environments
- metalotolerants: can survive high levels of dissolved heavy metals
- polyextremophiles: qualifies as an extremophile in more than one category
Many astrobiologists look at this long list of extremophiles as support for the notion that life must be a common feature on planets and moons throughout our Milky Way Galaxy and possibly on other galaxies as well. Consequently, the recent announcement from a NASA research team that it had found a bacterial strain that can grow by using arsenic, instead of phosphorus, solidified this presumption. It also elicited a near euphoric response from scientists and others wishing to explain life’s existence and diversity without invoking the handiwork of God.
Within minutes of NASA’s announcement, Wired’s science section ran the headline “NASA Unveils Arsenic Life Form.”1
NASA associate administrator for the Science Mission Directorate Ed Weiler declared, “The definition of life has just expanded.”2
Leader of the NASA research team Felisa Wolfe-Simon claimed, “We’ve cracked open the door for what’s possible for life in the universe. And that’s profound. What else might we find?”3
The discovery has been touted as evidence for a “second genesis” of life on Earth. Physicist Paul Davies, a member of the NASA research team, commented, “If life happened twice on one planet, it is sure to have happened on other planets around the universe.”4
A look at the team’s peer-reviewed paper reveals more subdued claims.5 The researchers did not discover a naturally occurring arsenic-based life-form. Rather, they cultured microbes from Lake Mono sediment in the lab and these microbes demonstrated some capability of absorbing arsenate under controlled chemical conditions. Mono Lake (see figure 1) is three times saltier than ocean water and highly alkaline. Its sediment is very arsenic rich and relatively phosphorus poor. Thus, the researchers already knew that any microbes existing in Mono Lake sediment would have to be arsenic tolerant. These microbes qualify as both alkalophiles and metalotolerants.

Figure 1: Mono Lake
Situated on the east side of the Sierra Nevada in central California, Mono Lake has a surface area of 69 square miles. Tufa towers like the one pictured above are calcium carbonate spires formed by the interaction of freshwater springs with the alkaline lake water.
Image credit: Mila Zinkova
Wolfe-Simon’s research team found a strain, dubbed GFAJ-1, capable of surviving and even growing and reproducing (albeit at much lower rates) in an artificial environment that was fed loads of arsenate but little or no phosphate. Analytical techniques performed by the team demonstrated that GFAJ-1, a member of the microbial family Halomonadaceae (see figure 2), was to some extent starting to use arsenate as a building block in place of phosphate.
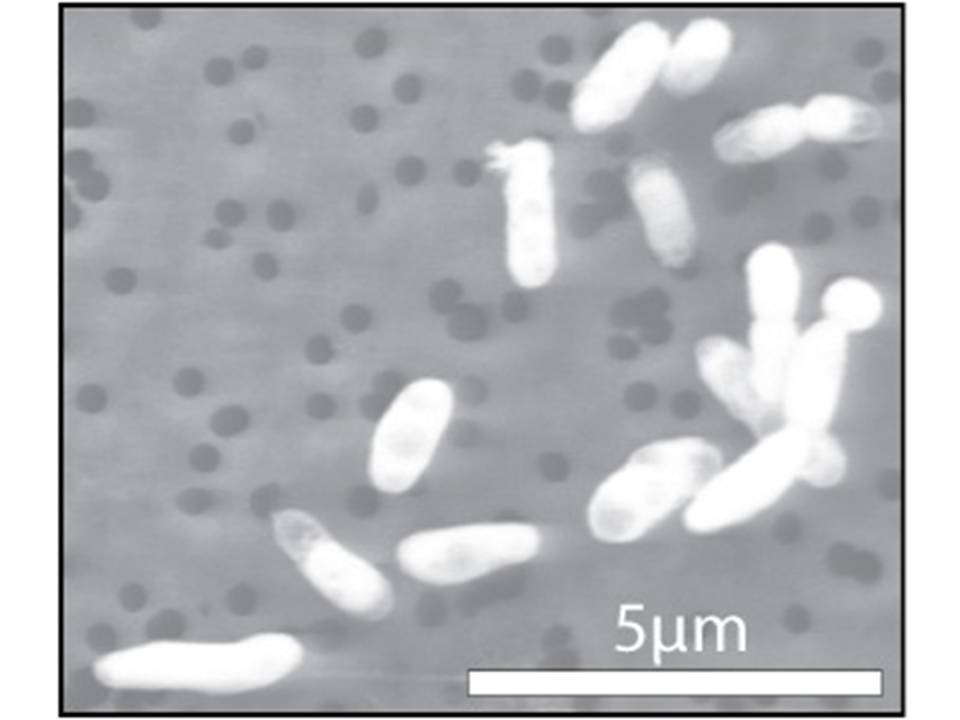
Figure 2: Halomonadaceae
This particular strain of Halomonadaceae, GFAJ-1, was grown in an environment with a normally occurring abundance of phosphorus. It did not incorporate any measureable arsenate into its DNA, RNA, or protein molecular structures.
Image credit: NASA
The achieved result is not all that surprising since arsenic sits right below phosphorus in the periodic table. Therefore, arsenic exhibits similar chemical properties with phosphorus. Without the availability of phosphorus, GFAJ-1 has no other alternative but to use arsenic.
One problem with arsenic, however, is that arsenate esters are not as stable as phosphate esters. In fact, the team noted that the GFAJ-1 strain that incorporated arsenate esters did not grow as fast as GFAJ-1 that did not. They also observed that the arsenate-incorporating GFAJ-1 did so only at a partial level. The “successful” strain did not replace all of its phosphate with arsenate. Rather, it tolerated some limited arsenate incorporation.
Bacteria can tolerate molecular instability much better than other life-forms thanks to their very short life spans and generation time scales that measure in minutes rather than years. In most cases, molecular instability would not register because it would take longer to develop than the bacteria’s average generation time. Furthermore, GFAJ-1 being both an alkalophile and metalotolerant likely would have within its molecular machinery efficient means for repairing DNA and RNA damage generated by its exposure and incorporation of arsenic.
The team did make clear that their arsenate-incorporating GFAJ-1 strain is still carbon-based life. Arsenic only replaced some of the phosphorus within GFAJ-1’s carbon-based molecules. They also made clear that arsenic is a vital poison. For many species of life, including human beings, arsenic plays a vital role. Too much arsenic will kill a human, but too little will also prove catastrophic. For optimal health, arsenic should make up one ten-millionth of a human’s body mass. For the research team’s laboratory-cultured strain of GFAJ-1, the arsenic quantity was pushed to a far higher level.
Has this altered strain of GFAJ-1 “greatly aided our search for extraterrestrial life” as many in the blogosphere now claim?6 The truth is that no naturally occurring environment will ever manifest the circumstances set up by the research team in their laboratory. In their experiment arsenic was made far more abundant than phosphorus. Within Earth’s crust arsenic measures 667 times less abundant than phosphorus. The cosmic abundance of phosphorus ranks 2,500 times greater than that of arsenic.
Another problem with suggesting that the altered GFAJ-1 greatly aids the search for extraterrestrial life is that its one thing for life to survive in an extreme environment, it is quite another for that life to originate in an extreme environment. Back in 2004, a geologist and biochemist published a paper explaining why the existence of extremophiles on Earth is irrelevant to the origin of life.7 As Fuz Rana and I point out in our book Origins of Life, as impossible as a naturalistic origin of life might be under the most benign natural conditions, it is orders of magnitude more difficult under the conditions in which extremophiles live.
We credit Wolfe-Simon’s team for making a remarkable discovery. They have proven just how resilient and tolerant some of Earth’s extremophiles can be. Such results testify of the Creator’s intellect and supernatural power in designing extremophilic life’s molecular machinery with the defense and repair mechanisms necessary to survive in extreme environments.
The psalmist declares, “How many are your works, O Lord! In wisdom you made them all; the earth is full of your creatures.”8 Evidently, the Creator was so motivated to fill Earth with as much life as his chosen laws of physics would permit that he created the fullest possible panoply of extremophiles. We humans are the direct beneficiaries of God’s extravagant creative efforts.
Endnotes
- Rachel Ehrenberg, “NASA Unveils Arsenic Life Form,” Wired, December 2, 2010, https://www.wired.com/wiredscience/2010/12/nasa-finds-arsenic-life-form/.
- “Discovery of ‘Arsenic-bug’ Expands Definition of Life,” Tony Phillips, ed.,Science@NASA, December 2, 2010,
https://science.nasa.gov/science-news/science-at-nasa/2010/02dec_monolake/. - Alasdair Wilkins, “Everything you need to know about NASA’s ‘completely new form of life,’”io9.com, https://io9.com/5704600/everything-you-need-to-know-about-nasas-new-life, accessed December 13, 2010.
- Richard A. Lovett, “NASA Life Discovery: New Bacteria Makes DNA With Arsenic,” National Geographic, December 2, 2010, https://news.nationalgeographic.com/news/2010/12/101202-nasa-announcement-arsenic-life-mono-lake-science-space/.
- Felisa Wolfe-Simon et al., “A Bacterium That Can Grow by Using Arsenic Instead of Phosphorus,” Science (December 2, 2010), published online in Science Express.
- Wilkins, https://io9.com/5704600/everything-you-need-to-know-about-nasas-new-life.
- H. James Cleaves II and John H. Chalmers, “Extremophiles May Be Irrelevant to the Origin of Life,” Astrobiology 4 (March 2004): 1–9.
- Psalm 104:24.





